Perceval PlusTM Lancelot
PercevalTM Plus
Since its first clinical use in 2007, the Perceval platform has continuously evolved, guided by real-world experience, clinical evidence and the needs of Surgeons. Today, Perceval represents a proven platform, designed to deliver reliable performance while supporting the patient’s tomorrow.
PERCEVAL platform is a revolution built through continuous evolution.
CC-MK-001525 B
Product Overview
YOUR CHOICE FOR today AND tomorrow





Durability you can trust
Concistrè et al., JCVM, 2023 – 1157 pts

Made to last: FREE tissue treatment
Rat subcutaneous implantations.
The subcutaneous tissue implantation in rat model is the gold standard to assess tissue propensity to mineralize in vivo, as it resembles the dystrophic mineralization occurring in human patients years after implantation.
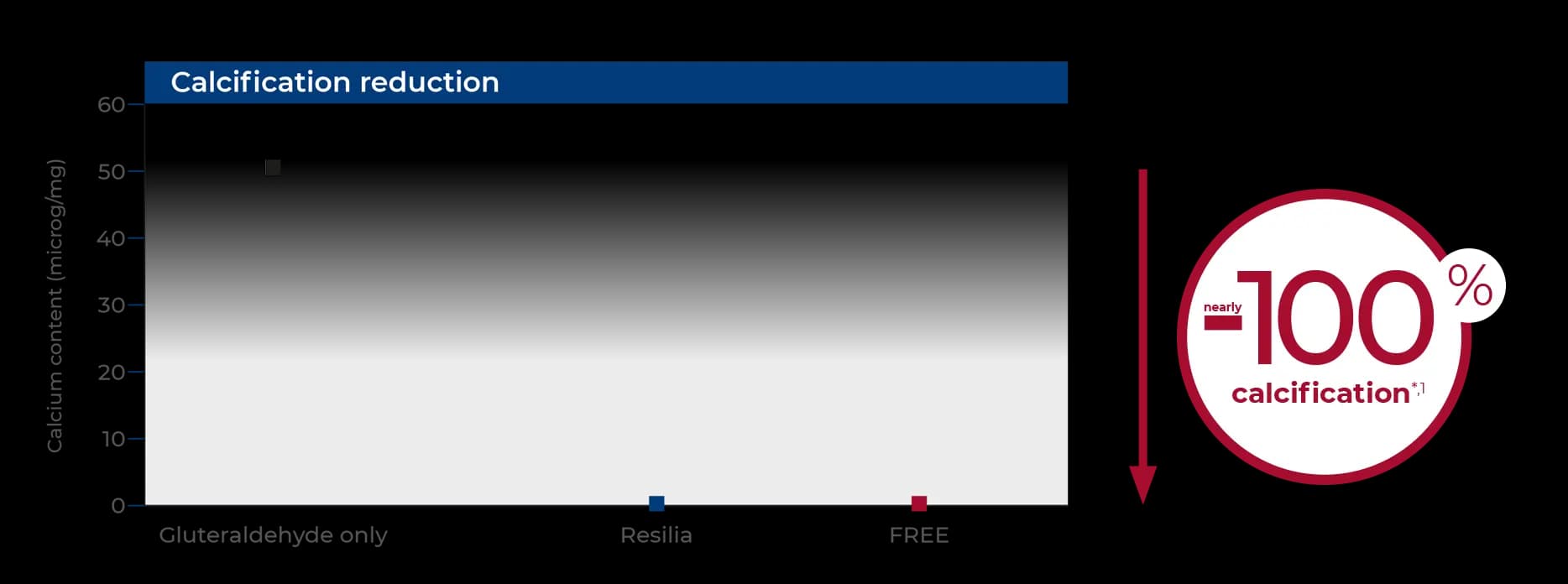
*vs control group
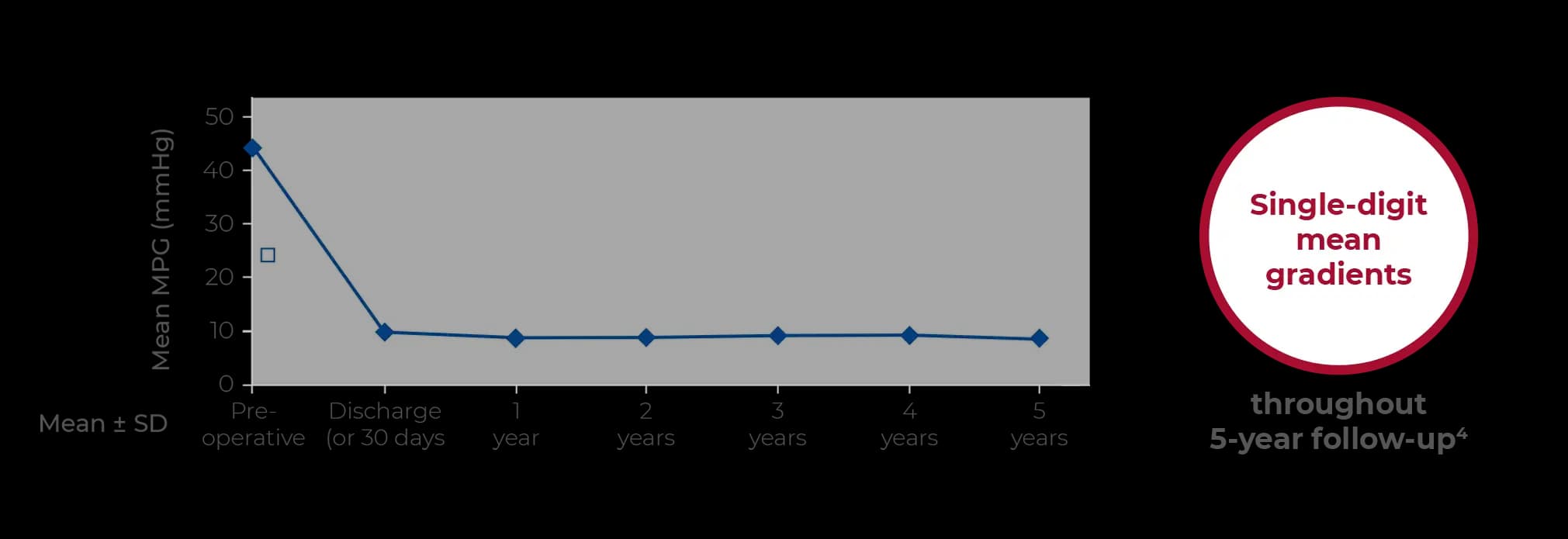
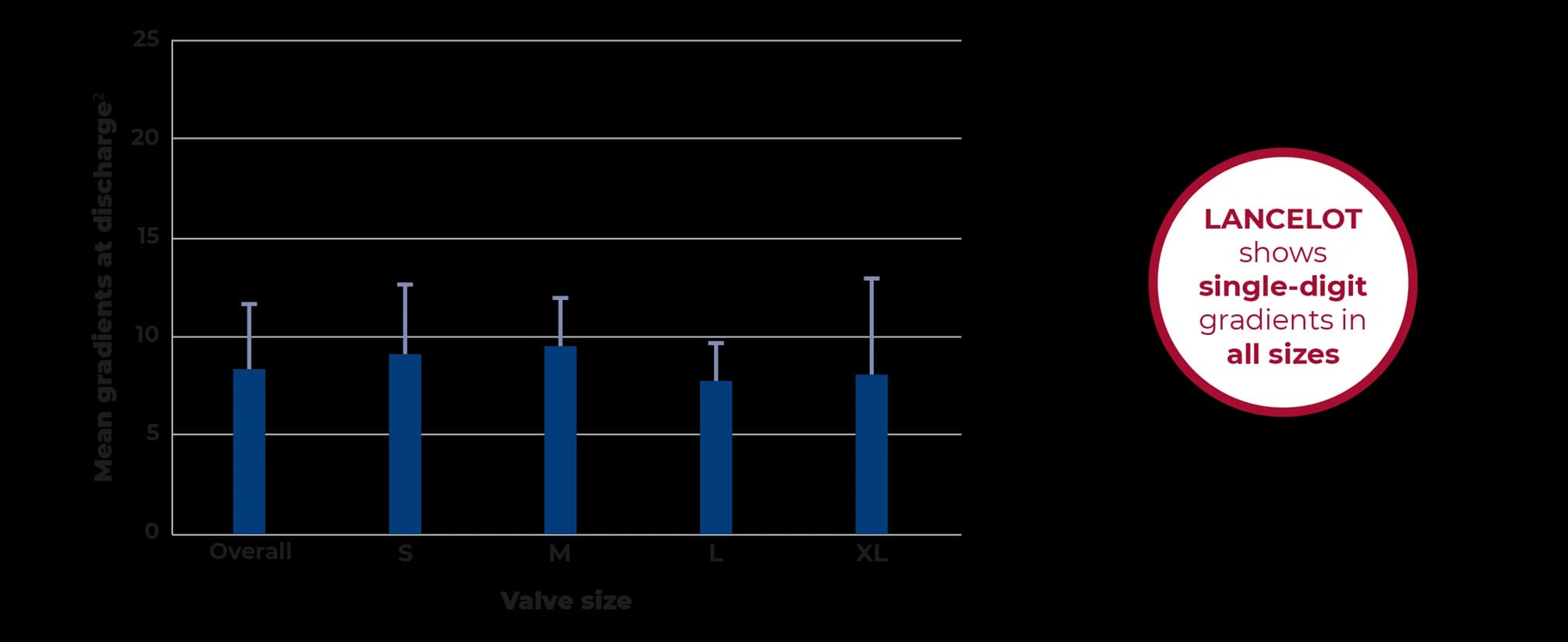
Designed for hemodynamics
The radial force at the inflow ring and the design of the superelastic stent allow for excellent hemodynamics with stable results over time.3,4
References
Abstract presented at SCTS Annual Meeting
2025
UZ Leuven experience – CORCYM data on file
European multicentre experience with the sutureless Perceval valve: clinical and haemodynamic outcomes up to 5 years in over 700 patients.
Eur J Cardiothorac Surg. 2015
Midterm outcomes with a sutureless aortic bioprosthesis in a prospective multicenter cohort study
J Thorac Cardiovasc Surg 2021
Safety Information
IMPORTANT SAFETY INFORMATION DOCUMENT
Manufactured by:
Corcym S.r.l.
Via Crescentino sn
13040 Saluggia (VC) Italy
Теl: +39 0161 1640001
Corcym S.r.l. previously Sorin Group Italia S.r.l.
Corcym Canada Corp.
5005 North Fraser Way Burnaby,
BC V5J 5M1 Canada
Tel: +604 412-5650
Corcym Canada Corp. previously LivaNova Canada Corp.
0123












